The Power of Fleet Management in Distributed Manufacturing

The pharmaceutical industry faces significant challenges in rapidly expanding production capacity to meet the needs of patients. Traditional centralized manufacturing models are increasingly seen as inflexible and slow to adapt to the dynamic demands of modern healthcare. This article proposes a regulatory framework for distributed manufacturing using the concept of fleet management to address these challenges.
Fleet management involves creating a network of standardized manufacturing units or equipment that can be efficiently managed and replicated across one or multiple sites. This approach draws inspiration from the principles of standardization that revolutionized other industries, such as automotive manufacturing.
The proposed approach offers significant benefits, including improved regulatory efficiency, reduced drug shortages, and enhanced environmental sustainability. This approach calls for collaboration between regulators and industry stakeholders to refine and implement this innovative framework, which will pave the way for a more resilient and responsive pharmaceutical manufacturing ecosystem.
Challenges
Pharmaceutical companies often encounter significant challenges when investing in processing equipment due to the industry’s stringent standards and need for customized production solutions. Collaboration with technology vendors frequently leads to the creation of bespoke systems designed to meet company-specific requirements. Although these tailored systems provide short-term benefits, they also introduce long-term complexities. Each system evolves uniquely through multiple iterations and adaptations to different production environments, resulting in diverse configurations that carry distinct risks, especially with respect to supply chain design and technology transfer. These specialized failure modes demand expert knowledge to manage, adding layers of complexity to the equipment’s life cycle. Furthermore, the lack of standardization in these systems presents significant challenges for the manufacturers and the regulators, who must invest additional time to assess each solution. This variability complicates production transfer between sites, raising operational costs and slowing down manufacturing processes. As the pharmaceutical industry progresses, there is a growing recognition of the need to move beyond traditional manufacturing approaches. The currently well-established centralized production model nonetheless has certain barriers that can lead to longer lead times, increased transportation costs, and supply chain vulnerabilities.
To address these challenges, the pharmaceutical industry is increasingly shifting toward a more agile manufacturing framework that combines the efficiency of large-scale production with the adaptability of smaller, distributed facilities. Regulatory agencies, including the European Medicines Agency (EMA) and the US Food and Drug Administration (FDA), are actively creating guidelines to facilitate this transition, labeling it as decentralized manufacturing (EMA) 1 or distributed manufacturing (FDA)2, 3. In this article, we will refer to it as “DM.”
Fleet
Central to this evolution is the concept of “fleet,” a DM operative model that represents a transformative approach to pharmaceutical production. Fleet is founded on the principles of standardization that gained prominence during the Industrial Revolution, emphasizing the creation of uniform systems that can be efficiently replicated and managed across multiple sites. This approach draws parallels with the automotive industry’s journey, where innovators like Ford revolutionized manufacturing through standardized processes that drastically reduced costs, enhanced quality, and facilitated knowledge sharing 4.
In the pharmaceutical field, similar advantages have been noted in technologies like fermentation units, freeze dryers, and autoclaves, where uniform design and operation have improved efficiency and minimized complexity. Fleet aims to push these principles further, advocating for a standardized methodology that transcends site-specific differences. This will help foster consistency and uniformity across all production facilities.
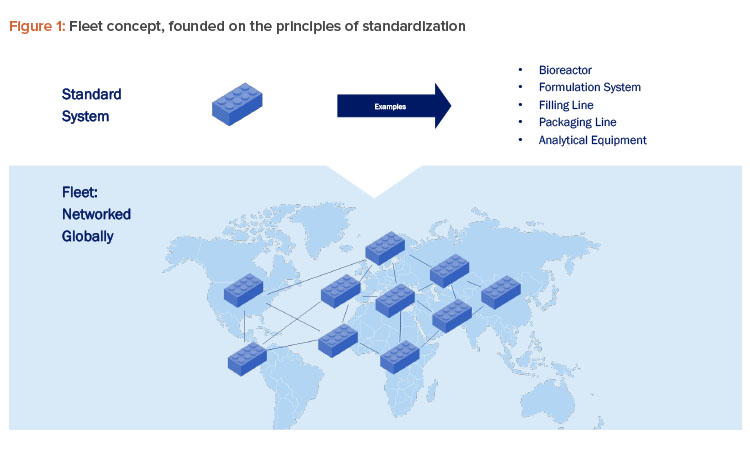
Implementing the fleet concept, illustrated in Figure 1, involves establishing rigorous controls and standardized procedures to ensure system equivalence across locations and throughout the life cycle. A key aspect is a central quality system within a network of sites to maintain product quality and regulatory compliance through standardized protocols (e.g., qualification/validation, calibration, maintenance). Developing a comprehensive oversight framework is essential for managing the entire life cycle of the fleet, including procurement, operation, and decommissioning.
This framework should encompass detailed documentation, regular audits, and continuous training for personnel. By integrating risk management strategies and leveraging real-time data analytics, organizations can proactively address potential issues. Fostering collaboration among stakeholders enhances communication and promotes best practices, ultimately leading to improved operational efficiency, reduced variability, and consistent delivery of high-quality products in the pharmaceutical industry.
This article outlines the core elements of the fleet approach. It focuses on its organizational structure, its key components, and the strategic benefits of efficient fleet management within the DM framework. Through an exploration of DM’s fundamental principles, we demonstrate how the fleet can boost production efficiency, mitigate drug shortages, and drive innovation within the pharmaceutical sector.
The primary definitions are summarized in Table 1. We aim to offer a thorough understanding of DM’s potential to revolutionize pharmaceutical manufacturing, ensuring adherence to regulatory standards while maintaining exceptional product quality. Additionally, it proposes a progressive regulatory framework that emphasizes current GMP (cGMP) compliance, safety, and traceability, highlighting the need for adaptable regulatory strategies to meet the dynamic demands of modern manufacturing.
| Term | Definition |
|---|---|
| Centralized Pharmaceutical Quality System (PQS) | A PQS that employs a grouping strategy or family approach to the specification, design, procurement, qualification, operation, equivalency, and life cycle management of the systems and equipment. |
| Equivalency | State in which manufacturing systems remain identical in critical aspects (e.g., specification, design, function, operation, and qualification approach) required for cGMP manufacturing execution. |
| Equivalent Manufacturing Systems | Manufacturing systems are defined as equivalent if they are identical in critical aspects (e.g., specification, design, function, operation and qualification approach) required for cGMP manufacturing execution to deliver robust process performance and consistent product quality over the life cycle. |
| Fleet | Group of equivalent systems that are constructed and installed to the same specifications across multiple manufacturing facilities across different locations, networked and operated by a central PQS. |
Background
The Regulatory Landscape and Its Challenges
Existing pharmaceutical regulatory standards are designed to ensure the safety, efficacy, and quality of medicinal products, established by various international regulatory bodies such as the US FDA and EMA. These standards encompass comprehensive guidelines that cover the entire life cycle of a pharmaceutical product, from research and development to manufacturing, marketing, and post-market surveillance.
Key components include cGMP, which enforces strict controls on production processes to maintain consistency and compliance with quality specifications. Additionally, the frameworks prioritize thorough clinical trials and rigorous approval processes to ensure that new drugs are safe and effective for their intended use. However, these regulations are primarily tailored to traditional manufacturing models, which can pose challenges when applied to the complexities of DM networks that aim to reach more patients efficiently.
Under the current regulatory framework, pharmaceutical manufacturers face stringent requirements when expanding or establishing new locations within a production fleet. This includes demonstrating bioequivalence, generating analytical comparability data, and validating methods and processes for each site. However, when manufacturing units can be proven equivalent in design, qualification, and operation, the necessity for duplicating extensive data for every new site may be reevaluated, as equivalency of the fleet members minimizes risks to drug product quality.
A significant limitation in the current system is the lack of a clear regulatory pathway for submitting and approving DM networks. Despite having a central PQS, existing guidelines 5 tend to treat each site and product as unique, without a defined framework for managing quality control and validating processes across multiple locations. This gap highlights the urgent need for updated regulatory guidance that outlines submission categories, strategies, and documentation requirements specific to DM. This is because the absence of such updates complicates ensuring the safety, efficacy, and quality of products manufactured through distributed systems.
The industry and health authority responses to the COVID-19 pandemic showcased the potential for innovative thinking and operational changes beyond traditional pharmaceutical models. During the pandemic, industry supply chains largely met their goals, and governments demonstrated a willingness to intervene to ensure the security of pharmaceutical supplies. These actions exemplify how regulators and industry collaborated to quickly enhance manufacturing capacity and effectively distribute medicinal products to the public. Drawing on these experiences, there is an opportunity to work jointly with health authorities to develop a DM regulatory framework that prioritizes global access of medicines and patient needs.
DM in the Pharmaceutical Industry
There is a growing trend toward the localization of supply chains and a push for regional or national self-reliance. This shift is driven by several factors, including changes in the globalization model, the diminishing relevance of global trade agreements, and an increased emphasis on sustainability—all elements that can affect industry supply chains. As a result, unpredictable shocks in the future are likely to reinforce the trend toward supply chain flexibility, and to develop faster product transfers to achieve higher adaptability and product availability.
Portfolio trends are driving significant shifts in the traditional pharmaceutical model, with one of the key factors being the rise in competition. As patents on existing drugs expire, biopharmaceutical companies increasingly focus on boosting manufacturing efficiency to reduce costs. With each new medicine aiming to surpass its predecessors, the emphasis on high-quality production processes continues to grow.
Furthermore, the increasing prevalence of complex biologics requires specialized approaches to manufacturing, storage, and handling, necessitating a precisely managed supply chain. In this context, achieving manufacturing excellence through innovation is essential to ensure adaptability, uphold quality standards, and meet cost-efficiency goals in response to these changing dynamics.
Challenges Establishing a Regulatory Framework for DM
Developing a regulatory framework for DM poses significant challenges in maintaining product quality and patient safety across multiple sites. A key issue is ensuring system equivalence when different manufacturing locations have varying processes and environmental conditions. To address this, robust criteria and advanced technologies like real-time monitoring and data analytics are recommended to standardize quality metrics and quickly identify deviations.
Establishing standardized protocols for equipment qualification, calibration, and validation, as well as implementing consistent training programs, will further support quality assurance. Effective collaboration between regulatory agencies and manufacturers is essential to create clear guidelines for assessing variability without compromising product quality.
Another challenge is creating a centralized PQS that ensures consistency and regulatory compliance throughout the DM network. This system requires harmonized protocols that account for the unique conditions of each site while maintaining uniform quality standards. Regulatory bodies must consider their inspection practices to determine whether to inspect sites individually or collectively, balancing thorough oversight with efficiency. Open communication and best practice sharing among sites will be crucial for the success of this centralized PQS, as it enables real-time monitoring and proactive adjustments across all locations.
Implementing new manufacturing processes within a DM network adds complexity for the initial unit, requiring careful planning and seamless integration to avoid disruptions; however, it ultimately streamlines operations at the network level and therefore to the additional fleet members. Future DM guidelines from regulatory agencies might focus on assessing and validating these changes to ensure they do not compromise product safety or efficacy.
Current data submission requirements also present challenges, as they can be time-consuming and costly. A more flexible, risk-based regulatory approach could be established that considers the specific conditions and performance of each fleet member together with the knowledge and understanding from the DM network’s fleets. This would streamline the process while maintaining high standards of quality and safety, allowing for more agile and cost-effective manufacturing expansion.
Proposed Framework for DM
The philosophy behind DM is the concept of “design one, build many” (copy/paste) and is operationalized using fleets. The fleet concept serves as a key mechanism to ensure the standardization of equivalent manufacturing systems across various sites, regardless of location, throughout the entire life cycle of the fleet. A central governing body is responsible for maintaining these standards across all stages of the fleet’s life cycle, ensuring uniformity and compliance across different facilities. This approach fosters continuous improvement, allowing for cross-network problem solving, knowledge sharing, and enhanced process robustness.
The fleet concept enables a more streamlined and accelerated qualification process, making ramp-ups faster and more efficient. A comprehensive fleet management system ensures that all fleets, regardless of their geographic location, adhere to established fleet standards. With a highly connected digital network, fleet performance is monitored and controlled in real or near real time, ensuring alignment and consistency across the entire DM network.
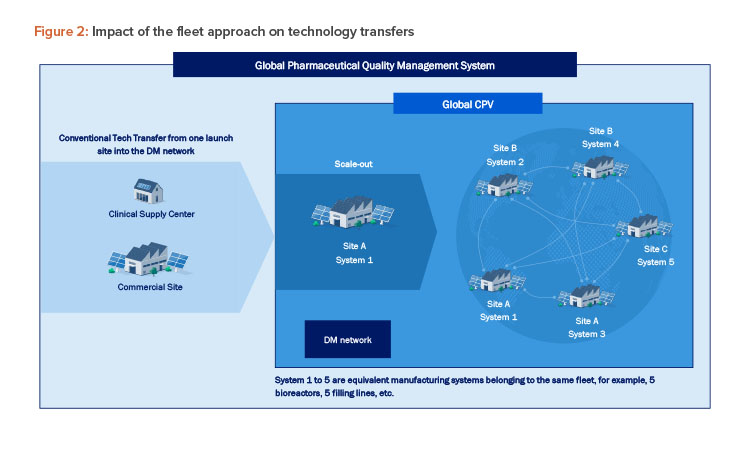
Establishing a fleet of equivalent manufacturing systems across multiple sites allows systems within a fleet to leverage data, knowledge, and understanding from other fleet members to expedite the risk-based approval and implementation of subsequent members. This reduces the need for extensive validations, stability studies, and comparability studies, as well as the frequency and scope of inspections, as health authorities can rely on the data and experience from other fleet members. The fleet approach simplifies the initial technology transfer process and ensures consistent operations across the DM network. New members can be seamlessly integrated into the network, as subsequent technology transfers are efficiently handled through the company’s PQS (see Figure 2), reducing the need for further regulatory reviews.
The systems within a fleet will benefit across various phases, including procurement, design, construction, qualification, validation, operation, ramp-up, and production. Table 1 highlights the potential benefits of fleet systems.
| Potential benefits of utilizing the fleet concept |
|---|
|
A Centralized PQS
The International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human use (ICH) “ICH Harmonised Tripartite Guideline Q10: Pharmaceutical Quality Systems” guidance 5 was introduced to bridge the gap between traditional cGMP and the evolving demands of modern PQS. ICH Q10 fosters a culture of innovation and quality by design (QbD), where companies can proactively address potential risks, optimize processes, and implement corrective and preventive actions (CAPAs) effectively. In doing so, companies not only meet compliance requirements but also reduced time-to-market and enhanced patient safety.
Several pharmaceutical companies already have a global quality management system or centralized PQS, which establishes standards and expectations that apply uniformly across all locations, guiding the development of any local procedures. A centralized PQS contains the four core pillars described in ICH Q10, which are essential for fleet management, supporting each stage of the product life cycle:
- Process performance and product quality monitoring system
- CAPA system
- Change management system
- Management review of process performance and product quality
Implementing fleets of standardized production systems across multiple sites requires a shift from local to global thinking. Each site must operate as part of a broader, interconnected network, aligning technically and culturally to ensure that actions taken at one site are informed by the performance of the entire fleet. Highly integrated process performance and product quality monitoring systems is essential to provide near-real-time visibility across all sites, enabling proactive tracking of trends, identification of deviations, and prompt responses to variations.
Effective coordination across a manufacturing fleet relies on a robust change management process with clear roles and accountability. Fleet-wide visibility into process performance allows for the early detection of risks and the implementation of global CAPAs instead of localized solutions. Consistent change management fosters continuous improvement, ensuring product stability, safety, and quality while promoting a unified approach across the network.
A well-integrated PQS is crucial for managing changes across sites. Leveraging PQS tools like annual product quality reviews and trend analysis for continued process verification (CPV) (US FDA terminology) or ongoing performance verification (OPV) (EMA terminology) helps identify variations that could indicate emerging risks. Strong governance, combined with structured change management and a focus on fleet-wide equivalency, ensures consistent product quality and safeguards the integrity of the manufacturing process across all locations.
The cGMP oversight requirements for a centralized PQS mirror those for overseeing multiple manufacturing sites, contract manufacturing organizations, and suppliers. The key difference lies in leveraging advanced digital technologies, which enable real-time monitoring and oversight of remote units, ensuring that quality standards are maintained across the board despite geographical separation.
The establishment of enterprise and cloud-based systems—such as a manufacturing execution system, laboratory information management system, change management or deviation tracking platforms—that can be accessed globally allows for centralized control and oversight. These systems will contain historical records for each member of the fleet, enabling comprehensive tracking and trend analysis across the entire network. This approach strengthens consistency in quality and compliance.
ICH Q10 specifies that the effectiveness of the PQS can typically be assessed during a regulatory inspection conducted at the manufacturing site. In the context of a fleet approach, the PQS could be inspected by health authorities as a single unified entity, irrespective of the number and geographical distribution of the fleet members. This inspection process would be based on a risk-based approach, considering the overall maturity and performance of the fleet.
The centralized PQS, along with data and information shared between the central organization and individual fleet members, would play a critical role in guiding this evaluation. The high level of connectivity between fleet members allows authorities to not only assess the performance of individual sites but also review the overarching fleet governance system.
This ensures that consistent standards, procedures, and quality controls are applied across all locations. By leveraging this centralized approach, the inspection process becomes more streamlined, enabling regulators to focus on system-wide compliance rather than conducting redundant inspections at each site. Furthermore, the ability to monitor performance metrics and procedures across the network ensures that inspections are more targeted, potentially reducing the time and resources needed to complete them. This leaner inspection process can lead to more efficient regulatory oversight while maintaining rigorous quality assurance standards across the DM network.
A targeted, streamlined inspection process could further support reliance among health authorities. For example, if a health authority inspects site A, which has specific equipment within a fleet used across multiple sites, then another health authority might not need to focus on that equipment during an inspection of site B. This approach could reduce redundant inspection efforts, promote resource efficiency, and enhance collaboration among health authorities by allowing them to rely on each other’s findings. This would ultimately contribute to a more cohesive and efficient regulatory oversight process.
Fleet Management
Fleet management is the process of defining and establishing a standard for a manufacturing system, replicating the standard across multiple sites, and maintaining the standard over the life cycle of the fleet. Fleet management begins when defining a consistent system design and user requirement specifications of a particular system to be replicated.
To ensure consistency and compliance across the DM network, a robust governance framework must be established to manage changes, as well as external factors such as environmental conditions, utility systems, personnel qualifications, and procedures. Managing the impact of surrounding areas needs to be taken into consideration during the definition of the border and interfaces of a specific fleet equipment.
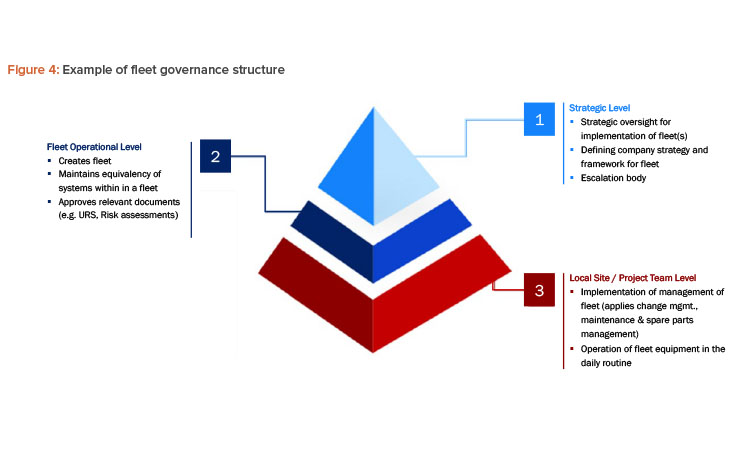
The governance framework should ensure the standardization and alignment of key aspects of the manufacturing environment to meet the operational needs of all fleet members. To achieve consistency across the fleet, a well-defined organizational structure that integrates both global and local roles is essential. This structure typically includes at least three tiers within the pharmaceutical company: strategic, fleet operation, and local site/project team levels, as depicted in Figure 4.
Strategic Level
This level functions as a strategic interface between the fleet management program and the company’s top management. It also serves as a liaison with parts of the manufacturing processes that follow the traditional approach rather than the fleet model.
Operational Level
The network group is the main body to maintain a fleet, especially maintaining the equivalency of equipment within a fleet. The global network group at this level could include:
- System owner
- A role per site owning a fleet equipment representing operations
- A role per site owning a fleet equipment representing quality
- Network, local subject matter expert (SME) representing a specific topic (e.g., regulatory, engineering) as required
Site representatives (operation and quality) act as an interface between the global network groups and the sites and should bring in the needs and requests from the sites into the global network group for discussion. They also have technical oversight for implementation of the fleet within their sites.
Local Site and Project Team Level
The local site and/or project teams are in close collaboration with the site representatives in the global network group. The local team operates fleet equipment or executes any work on fleet equipment.
Building a Global Fleet
Creating a standardized system design is a fundamental step toward achieving consistency in pharmaceutical manufacturing processes. This involves clearly defining essential components like user requirements and engineering specifications. Following this, the development of standardized documentation and validation practices, including templates for protocols, test plans, and reports, is crucial. Operational procedures are also aligned across the board, covering job instructions, process flows, training, and maintenance protocols. To ensure these standards are maintained consistently, a system owner is appointed to oversee the system’s life cycle across all locations, guaranteeing a unified approach.
After the standard design is established, it sets the stage for building a fleet of systems that strictly adhere to the specified criteria. This fleet consists of systems that are meticulously aligned in their design, functionality, and core processes, allowing only minimal variations. A System Owner oversees this fleet, ensuring that all systems perform cohesively, maintaining consistent manufacturing operations and product quality. This coordinated management approach ensures that the organization’s fleet operates as a unified entity, supporting reliable production outcomes.
Fleet qualification employs a science-driven, risk-based strategy to streamline qualification activities. Once the first system in the fleet is qualified, data and knowledge from this initial effort can be efficiently utilized to expedite the qualification of subsequent systems, reducing test documentation preparation, testing, and overall timelines. Performance metrics, tied to critical process parameters (CPPs) and critical quality attributes (CQAs), guide the continuous monitoring of fleet performance. Additionally, maintaining a fleet master plan as a dynamic document helps track fleet status, equipment details, and system boundaries. Regular updates to this living document ensure that any new equipment installations or system retirements are accurately reflected, keeping the fleet’s information up to date and fostering continuous improvement.
By establishing a network of manufacturing sites equipped with standardized processes and equipment, companies can swiftly adjust production locations to meet unforeseen demand spikes or local disruptions.
Change Request Process
To maintain the equivalency of a specific fleet, a stringent change control process is of utmost importance. The impact of each change must be analyzed accurately for the equivalency of the fleet members. A deep knowledge of the equipment, materials and processes are fundamental to making decisions upon impact of changes. If a change influences the equivalency, the change must be implemented on each piece of equipment within the fleet to maintain the equivalency. Therefore, the benefit of a change versus the requirements to implement in the fleet must be evaluated very carefully.
The global network group will be responsible for fulfilling this task. It is elementary that the people representing the sites in this group are well integrated in their sites to make sure that any events at a specific site impacting fleet equipment (e.g., changes, deviations, shutdowns, maintenance, internal audits) are reported to them in a timely manner, so that they can escalate it into the global network group for further evaluation or assessment. Any decisions impacting the equivalency of equipment within a fleet should be made within the global network group, balancing out pros and cons and impacts for each site. Specific topics need to be escalated to the next level, in case strategic decisions are required.
Independently of maintaining the equivalency within a fleet, continuous improvement is still an important aspect within the life cycle of this equipment. Continuous improvement should be performed at the supplier of fleet equipment as well as within a company using fleet equipment. The fact that there is more than just one machine within a fleet offers the possibility to gather more information and data about equipment and processes that are due to the equivalency directly comparable. Based on the availability of comparable data, improvements can be identified and implemented more quickly and efficiently to create more robust processes.
Knowledge Management (KM) in a Fleet
KM is crucial for maintaining fleet standards, ensuring equivalency, and enhancing overall performance across the fleet. ICH Q10 defines KM as a systematic approach to acquiring, analyzing, storing, and sharing information related to products, manufacturing processes, and components. In the context of a fleet, this involves applying and managing knowledge related to both the fleet and its individual members. Combined with the CPV/OPV program, KM plays a key role in driving performance improvements and ensuring operational consistency.
Building and Sharing Knowledge
A comprehensive fleet knowledge base should be developed to capture both explicit data and tacit knowledge from global network groups. This information must be systematically collected, organized, and shared across the fleet organization to ensure it reaches the right teams at the right time. The aim is to equip teams with the necessary insights to operate, support, and manage fleet members and their processes effectively. Advanced digital tools and connectivity enhance the management and flow of this data, creating a robust framework for knowledge sharing and decision-making.
Integrating advanced data analytics within the fleet transforms data into actionable knowledge, enabling strategies like early troubleshooting, impact analysis, and predictive maintenance to prevent equipment failures. This analytical approach drives continual improvement, optimizing fleet performance, training, and operations. Knowledge sharing across the network also extends to external stakeholders, such as quality professionals and regulators, fostering a collaborative culture and informed decision-making throughout the fleet organization.
Operational support across the fleet is seamlessly integrated through standardized processes, equipment, standard operating procedures (SOPs), and work instructions. This consistency allows colleagues and subject matter experts to quickly step in when issues arise. Digital tools like virtual reality and augmented reality can enhance interactive support experiences. When breakdowns occur, local teams provide the first response, while the global network offers additional support, ensuring comprehensive solutions that are consistently applied throughout the fleet.
Training
Standardized training across the fleet and the DM network ensures that all personnel follow the same protocols, assessments, and quality standards, equipping staff with consistent skills and knowledge. This unified approach enables effective support regardless of location and promotes seamless cooperation among team members, maintaining high-quality operations across the network. Specific roles and responsibilities are clearly defined, with dedicated training programs tailored to each role, continuously reviewed, and updated to reflect new developments and best practices. This standardized approach ensures that personnel adhere to consistent procedures and also facilitates efficient cross-site collaboration, enabling teams to provide effective support and adapt quickly to challenges.
CPV/OPV and Fleet Evaluation
Every pharmaceutical product requires a CPV or OPV program to confirm that the manufacturing process remains under control during commercial production. Typically aligned with the PQS, these programs use statistical tools to analyze CQAs and CPPs. When fleet members are digitally connected, CPV/OPV can be conducted across the network, enhancing the ability to monitor and improve the entire fleet’s performance.
Automated data collection and analysis provide real-time and near-real-time insights into both individual fleet member performance and overall fleet operations through a cloud-based system that integrates data from multiple sources. This setup enables continuous monitoring of product quality, equipment functionality, and fleet consistency. It also facilitates quick responses to deviations, implementation of global CAPAs, and initiation of improvement measures to maintain the fleet’s robustness.
The global availability of data from all fleet members is a critical factor in leveraging validation activities across the fleet, making the fleet management system uniquely effective. This comprehensive data access allows for better-informed decision-making and enhances the capability to maintain uniform standards and continuous improvement across all manufacturing sites.
Life Cycle Management
The management of fleet members within the DM network requires a comprehensive strategy to ensure the equivalence of manufacturing systems and equipment throughout their entire life cycle within a manufacturing network. One of the main challenges is overseeing equipment that may remain in use for decades, requiring meticulous maintenance, updates, and compliance checks to align with evolving standards and technologies.
The “design one, build many” approach offers a solution by promoting the use of standardized equipment across sites, which can streamline operations and reduce variability. Although this concept isn’t entirely new, what distinguishes the fleet management concept is its extension beyond the initial handover from project to operations, ensuring that standardization is maintained throughout the entire life cycle of the equipment. In pharmaceutical manufacturing, equipment life cycles often extend beyond a decade, during which evolving production needs or technological advancements may require additional equipment purchases. It’s crucial to have a long-term plan to maintain equipment equivalency, whether through upgrades to existing units or the introduction of a new fleet.
This approach poses challenges for pharmaceutical equipment suppliers, as they must strike a balance between providing versatile “off-the-shelf” solutions and offering highly customized options. Prioritizing the development of next-generation models over frequent modifications to standardized equipment may be more efficient. Effective fleet management, however, requires a strong partnership between suppliers and the pharmaceutical industry to ensure consistent performance, compliance, and operational efficiency across the entire network.
Effective management of supply chain quality is essential for the successful operation of manufacturing fleets, ensuring consistent quality and availability of critical raw materials such as drug substance precursors, single-use technologies, and assay reagents. A reliable network is key to maintaining equivalency across the fleet, supporting the broader manufacturing strategy, and ensuring that data from different locations are comparable.
Although cGMP facilities rigorously test incoming materials to verify their identity, purity, and quality, DM faces added complexities due to its reliance on local suppliers, which must be carefully managed to maintain consistency. These challenges could be addressed by the adoption of advanced analytical techniques, such as process analytical technology (PAT), real-time monitoring and control technologies, and strengthened supplier qualification processes. Effective communication with suppliers and solid contingency planning are also crucial to mitigate potential disruptions and uphold supply chain integrity.
A robust PQS is crucial for effectively leveraging the tools outlined in ICH Q12: Technical and Regulatory Considerations for Pharmaceutical Product Life Cycle Management 6 to manage the integration of new fleet members. Successful implementation of ICH Q12 tools necessitates a well-organized industry framework that draws on existing knowledge, scientific principles, and innovation. By employing the tools and enablers of ICH Q12, organizations can enhance fleet management throughout the product life cycle through a risk-based approach that specifies the appropriate level of regulatory oversight.
This proactive strategy, which includes post-approval change management principles, allows for better anticipation of future changes and their related reporting requirements, ultimately streamlining the regulatory process. Furthermore, a robust PQS, combined with effective KM and proactive quality risk management practices, can significantly improve operational flexibility, enabling the manufacturing process to adapt to changes while maintaining quality. This approach also ensures supply continuity by minimizing the risk of disruptions, thereby guaranteeing consistent product availability.
Additionally, fostering an innovative culture becomes more attainable as a well-managed PQS facilitates the effective integration of new fleet members, ensuring compliance and responsiveness to market demands throughout their life cycle. Aligning with ICH Q12 principles, where structured KM supports life cycle management, further enhances proactive change management and drives continuous improvement in the fleet operating model.
The fleet concept simplifies regulatory processes by standardizing data and procedures across multiple sites, reducing societal costs through streamlined production, decreased regulatory burdens, and improved drug supply reliability, ultimately benefiting all stakeholders involved.
Benefits of DM
DM manufacturing offers significant advantages, including enhanced patient safety, greater reliability and consistency in supply, speed in delivering medicines, improved compliance, and reduced societal costs. Enhanced patient safety is a core benefit of DM, made possible by the ability to share and leverage data, lessons learned, and best practices across multiple sites. This collaborative approach allows for broader perspectives, more comprehensive data analysis, and improved safety and performance across the network.
Drug shortages have become a pressing concern in the pharmaceutical industry, and DM offers a significant advantage in addressing this issue. One of the key benefits of the fleet concept is the enhanced reliability and responsiveness in patient supply. By establishing a network of manufacturing sites equipped with standardized processes and equipment, companies can swiftly adjust production locations to meet unforeseen demand spikes or local disruptions.
This interconnected approach bolsters supply chain robustness by creating “interchangeable sites” that can quickly adapt to changes in demand or supply challenges. Additionally, the capacity to share data and experiences across these sites further enhances overall performance and reliability. This collaboration not only facilitates a smoother rollout of new technologies but also helps to mitigate the risk of stockouts during emergencies, ensuring that patients have continuous access to essential medications.
DM fosters innovation and accelerates the delivery of medicines to patients by enabling a more agile and scalable production approach. Through the incremental rollout of fleet members across the network, continuous improvements can be applied in real time, minimizing trial and error and optimizing processes for smoother multisite implementation.
As discussed earlier, this approach not only enhances the overall efficiency of the network, but it also supports faster technology transfers between sites, which boosts flexibility and ensures a coordinated strategy across the entire fleet. As a result, the need for extensive comparability studies is significantly reduced, streamlining regulatory approvals and shortening the time-to-market for new medicines, ultimately improving patient access to life-saving treatments.
Another benefit of DM is its potential to enhance environmental sustainability within the pharmaceutical industry. By localizing production closer to end users, this approach significantly reduces the carbon footprint associated with long-distance transportation of goods. Additionally, DM often allows for the use of renewable energy sources at localized sites, further minimizing environmental impact.
Furthermore, the decentralized nature of DM fosters innovation in sustainable practices, as each site can implement localized strategies for waste reduction, recycling, and energy efficiency that align with community goals. This alignment not only supports corporate sustainability initiatives but also enhances corporate social responsibility, ultimately contributing to a more sustainable and resilient supply chain.
Finally, health authorities benefit from DM through reduced inspection demands, streamlined review processes, and a more robust supply chain that lowers the risk of stockouts. The ability to quickly adapt manufacturing to meet demand, scale out production processes, and enhance process reproducibility with replicated equivalent manufacturing systems supports regional production and improves patient access to essential medicinal products.
Conclusion
In this article, we outlined a comprehensive framework for the fleet concept as an operational model for DM. By focusing on the principles of scaling out and the copy/paste approach, we highlight how these core elements can significantly enhance compliance and minimize variability in the production of medicines. This alignment between industry practices and health authority objectives ensures the delivery of safe and effective drugs, whether they are new candidates in clinical trials or established therapies. The fleet concept simplifies regulatory processes by standardizing data and procedures across multiple sites, reducing societal costs through streamlined production, decreased regulatory burdens, and improved drug supply reliability, ultimately benefiting all stakeholders involved.
The flexibility inherent in this framework makes it also suitable for emerging modalities. By adopting the approaches presented in this article, manufacturers can significantly reduce waste and optimize resource efficiency while creating a production system that is agile and responsive to fluctuations in demand. This adaptability not only enhances inventory management but also ensures the consistent availability of critical products across global markets.
By fostering collaboration between regulators and industry stakeholders, we can work toward establishing a clear and robust regulatory environment that facilitates the implementation of these innovative manufacturing models. We have previously had informal yet constructive discussions with regulators from the US FDA and EMA about creating a suitable regulatory environment for the fleet concept, and both parties expressed support for continuing this dialogue.
We encourage regulators to keep engaging with industry sponsors to further refine and implement the ideas presented in this article. A well-defined regulatory framework that embraces the principles of the fleet concept will be crucial for advancing flexible, on-demand small or large volume production solutions, ultimately leading to a more efficient and responsive healthcare delivery system on a global scale.







