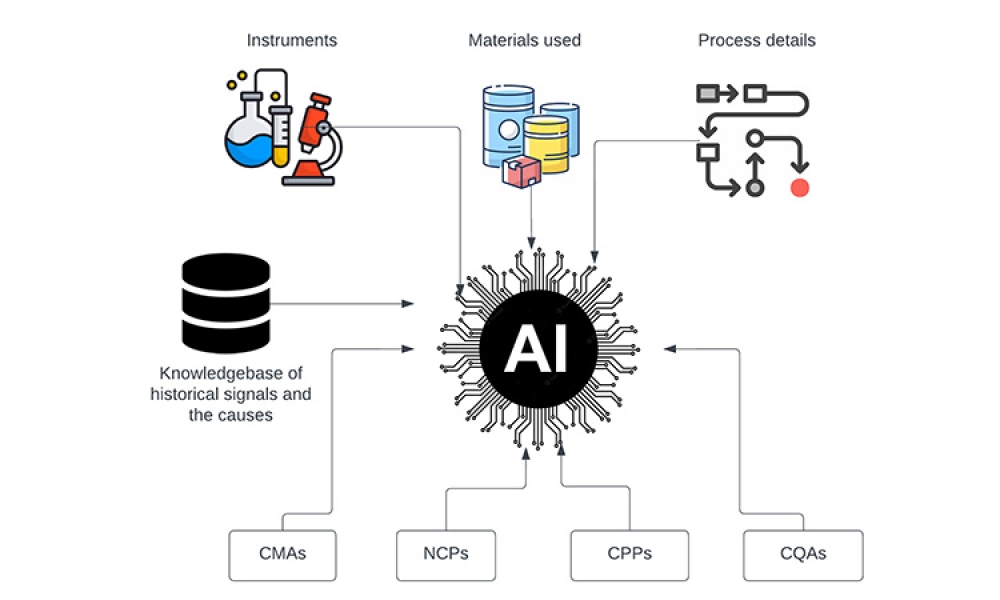
Validation is creating an evidence trail to show that an action, method, or system leads to a consistent and reproducible result. Validation is the collection and evaluation of data from the process design stage through commercial production, which establishes scientific evidence that a process or components of a process can consistently deliver a quality product. Process validation involves a series of activities taking place over the lifecycle of the product and process.
Guidance Documents
Data Integrity (10)
- GAMP Good Practice Guide: GxP Process Control Systems 2nd Edition
- GAMP Guide: Records & Data Integrity
- GAMP RDI Good Practice Guide: Data Integrity - Key Concepts
- ISPE GAMP RDI Good Practice GAMP Good Practice Guide: Data Integrity - Manufacturing Records
- GAMP Good Practice Guide: Calibration Management 2nd Edition
- GAMP Good Practice Guide: Operation of GxP Computerized Systems
- GAMP Good Practice Guide: Enabling Innovation
- GAMP Good Practice Guide: Regulated Mobile Applications
- GAMP Good Practice Guide: IT Infrastructure Control & Compliance 2nd Edition
- GAMP Good Practice Guide: GxP Compliant Laboratory Computerized Systems 2nd Edition
GAMP® (11)
- GAMP Good Practice Guide: Computerized GCP Systems & Data 2nd Edition
- GAMP Good Practice Guide: GxP Process Control Systems 2nd Edition
- GAMP Guide: Records & Data Integrity
- GAMP RDI Good Practice Guide: Data Integrity - Key Concepts
- ISPE GAMP RDI Good Practice GAMP Good Practice Guide: Data Integrity - Manufacturing Records
- GAMP Good Practice Guide: Calibration Management 2nd Edition
- GAMP Good Practice Guide: Operation of GxP Computerized Systems
- GAMP Good Practice Guide: Enabling Innovation
- GAMP Good Practice Guide: Regulated Mobile Applications
- GAMP Good Practice Guide: IT Infrastructure Control & Compliance 2nd Edition
- GAMP Good Practice Guide: GxP Compliant Laboratory Computerized Systems 2nd Edition
Lifecycle Management (1)
Microbiological & Viral Contamination Control (1)
Quality Assurance (1)
Validation (13)
- GAMP Good Practice Guide: Computerized GCP Systems & Data 2nd Edition
- Guide: Cleaning Validation Lifecycle - Applications, Methods, & Controls
- GAMP Good Practice Guide: GxP Process Control Systems 2nd Edition
- GAMP Guide: Records & Data Integrity
- GAMP RDI Good Practice Guide: Data Integrity - Key Concepts
- ISPE GAMP RDI Good Practice GAMP Good Practice Guide: Data Integrity - Manufacturing Records
- GAMP Good Practice Guide: Calibration Management 2nd Edition
- Good Practice Guide: Process Validation
- GAMP Good Practice Guide: Operation of GxP Computerized Systems
- GAMP Good Practice Guide: Enabling Innovation
- GAMP Good Practice Guide: Regulated Mobile Applications
- GAMP Good Practice Guide: IT Infrastructure Control & Compliance 2nd Edition
- GAMP Good Practice Guide: GxP Compliant Laboratory Computerized Systems 2nd Edition
Community Discussions
Community Discussions
Apr 16, 2025
Information Systems
Regulatory
Advanced Manufacturing
Artificial Intelligence
Apr 08, 2025
Validation
Mar 28, 2025
Information Systems
Regulatory
Advanced Manufacturing
Artificial Intelligence
Feb 03, 2025
Jan 27, 2025
Dec 04, 2024
GAMP®
Lifecycle Management
Oct 31, 2024
Regulatory
Quality
Good Manufacturing Practice
Sustainable Facilities, HVAC, & Controlled Environments
Pharmaceutical Engineering Magazine Articles
Webinars
White Papers
March / April 2024
Navigating the Asia Pacific Pharmaceutical Landscape for Global Impact Cover: The Asia Pacific…
Stage 3 Process Validation: Applying Continued Process Verification Expectations
This discussion paper proposes ideas for answering the questions “How is Stage 3 monitoring and…

Pharmaceutical Job Board
Videos
iSpeak Blog Posts
Featured Conferences

Professional Development Training
Requirements for Computerized Systems Validation and Compliance
This online course describes regulatory requirements and expectations regarding the validation and compliance of computerized systems used in the manufacture of pharmaceuticals, biologicals, and medical devices. It does not cover the detailed requirements of 21 CFR Part 11, except for the requirement that systems be validated. Even though it draws upon medical device guidance, it is not intended to cover all the requirements of producing software that subsequently becomes part of a medical device.
Commissioning and Qualification Training Course
Worldwide Regulatory expectations and guidance as led by FDA and the EU have stated that all Pharmaceutical Quality Systems should apply a QRM (Quality Risk Management) approach. Through interactive workshops, this course will explain and apply the science and risk-based approach to integrated lifecycle Commissioning & Qualification by conducting verification of systems, equipment and facilities in accordance with the recently issued 2nd Edition Guide, ICH documents Q8 (R2), Q9, and Q10, current Regulatory Guidance, industry best practices, and ASTM E2500.
Process Validation Training Course
This training course is relevant to individuals working throughout the pharmaceutical product lifecycle in development, manufacturing, quality, and many other roles involved in validation of products and processes. It will help you integrate and link the science and risk-based lifecycle approach for Process Validation to your overall Pharmaceutical Quality System.
Pharmaceutical Technology Transfer Training Course
This training course on Pharmaceutical Technology Transfer identifies criteria for successful technology transfer and provides ‘how to’ examples which can be individually tailored, depending on the type and scope of transfer.
Process Validation Biotechnology Manufacturing Training Course
This training course is designed to provide a clear understanding of the regulatory, scientific, and engineering tools required to successfully develop and validate bioprocesses. In addition, the course identifies the long list of activities required to validate biopharmaceutical processes.